By Sonia MR Vasconcelos*,**, Adriana B Graça**, Christiane C Santos***, Karina A Rocha*, Maria Júlia M Antunes*, Mariana D Ribeiro* and Marlise Pedrotti*
The COVID-19 Pandemic: More than a year ago…
The last day of the year 2019 marked the official start of a major change on the planet, which “… turned the world upside down. Everything was impacted. How we live and interact with each other, how we work and communicate, how we move… Every aspect of our lives has been affected. ”1 On December 31, 2019, the World Health Organization (WHO) was informed on the occurrence of atypical cases of pneumonia in the city of Wuhan, China.2 The spread of the outbreak would have started in a seafood and live animals’ market, a well-known hypothesis. However, to this day, the WHO still cannot confirm the real origin, according to its most recent report.
The causative agent of the outbreak identified in Wuhan was a novel coronavirus6, which would later be named SARS-CoV-2, by the International Committee on Taxonomy of Viruses.7,8 On January 30, 2020, the WHO declared this outbreak as a Public Health Emergency of International Importance – in the same month that the first death associated with the virus was recorded in China.7 Weeks later, on February 11, 2020, the disease caused by the novel coronavirus was named COVID-19 by the WHO – in reference to the type of virus and the year the epidemic started.7
In Brazil, on February 6, 2020, Law 13,9792 was published in the Diário Oficial da União with “… measures to deal with the public health emergency of international importance resulting from the coronavirus responsible for the 2019 outbreak.”2 When enacting the Law sanctioned by the Presidency of the Republic, the National Congress determined that, “to face the public health emergency of international importance resulting from the coronavirus”, a series of measures would be necessary. The text of the Law includes “isolation”, “quarantine”, “compulsory medical examinations” and “exceptional and temporary restriction on entering and leaving the country…”, among several other actions.2 Weeks later, in February 26, the first death by COVID-19 was confirmed by the Ministry of Health.
With the rise in cases in several countries, COVID-19 was characterized by the WHO as a pandemic on March 11, 2020. At that time, there was a natural uncertainty about the scale of the problem. The WHO did not yet have a case record for 81 countries and, for another 57, the record was ten cases or less. However, the Organization warned the world, that same day, of the more than 118,000 cases of COVID- 19 in more than 100 countries, with over four thousand deaths already registered. The WHO described COVID-19 as:
…an infectious disease caused by the novel coronavirus (SARS-CoV-2) and its main symptoms are fever, tiredness and dry cough. Some patients may experience pain, nasal congestion, headache, conjunctivitis, sore throat, diarrhea, loss of taste or smell, skin rash or discoloration of the fingers or toes. These symptoms are usually mild and begin gradually. Some people are infected but have only very mild symptoms. Most people (about 80%) recover from the disease without needing hospital treatment. One in six people infected with COVID-19 becomes seriously ill and has difficulty breathing. Elderly people and those with other health conditions like high blood pressure, heart and lung problems, diabetes, or cancer, are at greater risk of becoming seriously ill. However, anyone can get COVID-19 and become seriously ill.3
Since the international recognition that the scenario we had was a pandemic, COVID-19 has gained an overwhelming proportion. According to the WHO, more than 130,000,000 cases of COVID-19 and more than 2,800,000 deaths from the disease were confirmed worldwide by April 4, 2021. The COVID-19 cases were spread over almost the whole planet. Fortunately, at the moment when we talk about this problem, several countries have already started vaccination, such as China, the United Kingdom, the United States and several others, including Brazil. In Brazil, it is important to highlight the pioneering spirit of the Butantan Institute and the Oswaldo Cruz Foundation (FIOCRUZ).
Besides the vaccines already approved in several countries, up to January 6, 2021, “there are 63 candidates in the clinical evaluation in humans, and 172 candidate vaccines in the preclinical trial phase”.4 These figures reflect an intense and fast research activity on COVID-19, involving human beings, considering the period of less than a year since the official declaration of the pandemic by the WHO.
Research in humans in the pandemic – clinical trials in the spotlight
According to data from the Global Data report5, the number of clinical trials on COVID-19 increased by 639%. The report indicates that 549 clinical trials related to the disease were recorded in early April 2020. By January 2021, there were already 4,000 studies registered.
In this pandemic scenario, there is a type of study that has been gaining attention in the publication system and in the media, the so-called “human challenge trials”. Unlike conventional clinical trials, in challenge studies, “…volunteers are typically randomized to receive the vaccine being investigated or a control (a proven vaccine, an experimental vaccine, or a placebo).”6 These research participants, usually young and healthy, are then intentionally infected with the virus, in conditions controlled by the research team, rather than waiting for natural infection, as occurs in conventional clinical studies.
This type of study invites us to reflect on several ethical issues, which have attracted the attention of researchers in the clinical fields, in research ethics/bioethics and, also, from a portion of the public, in the United Kingdom, for example. Before addressing some of the ethical concerns, it is worth mentioning that, when it comes to the public in this pandemic scenario, there are two important phenomena. On the one hand, there is an enormous challenge related to the flow of public disinformation, alerted by the United Nations and the WHO in the first months of the pandemic. On the other hand, the possibilities for the public to become familiar with scientific information on public health and on COVID-19, although the understanding of this information cannot be taken for granted.
In the case of clinical trials, the public has been increasingly exposed to information, for example, which includes mention of the phases that make up these studies, by the WHO itself and other health organizations, by scientists, health authorities and the media. The Pan American Health Organization (PAHO), for example, released a few months ago, “Frequently asked questions about COVID-19 candidate vaccines and access mechanisms”, one of several information documents, in Portuguese.4 As described in this fact sheet, in the case of “vaccine candidates”, the “evaluation … undergoes different phases (preclinical and clinical) until a vaccine receives regulatory approval. The objective of this entire process is to ensure a safe and efficacious vaccine…”4
Information on the phases of clinical trials is also easily accessible, for example, on the websites of FIOCRUZ25 and Instituto Butantan:
Phase I: it is the first study to be carried out in humans and its main goal is to demonstrate the safety of the vaccine. Phase II: aims to establish its immunogenicity. Phase III: it is the last study phase before obtaining the health registration and aims to demonstrate its effectiveness. Only after completing the phase III and obtaining approval by the regulatory authority, the new vaccine can be made available to the population.7
Human challenge trials – debate on ethical issues reinvigorated
Human challenge trails rare, as compared to conventional clinical trials, which naturally reflects a more restricted space of information accessible to the public, such as related documents. In terms of scientific relevance, these studies may be important for conducting some of the conventional clinical studies themselves. In a report by a WHO Working Group, published in the journal Vaccine in January 2021, where several scientific articles were cited, it is considered that
SARS-CoV-2 challenge studies could add value to other types of vaccine research by enabling (a) accurate assessment of asymptomatic infection, (b) more rapid and standardized testing of multiple vaccine candidates, and (c) testing vaccines in contexts where there is little continuing transmission…8
In these studies, the selection of young and healthy volunteers, in addition to the protocols to monitor and care for these research participants after deliberate infection, are among the characteristics that, for some researchers, make this type of study defensible, especially in the COVID-19 pandemic.
However, research involving human beings and ethical issues are intertwined, which is even more evident for this type of study. For Professor Joerg Hasford, President of the Association of Medical Ethics Committees in Germany:
The concept of a challenge trial is that all participants are first vaccinated with placebo or with the test vaccine and then get intentionally exposed with a sufficiently high dose of SARS-CoV-2, so that all participants can be considered infected. As all participants have been exposed, the efficacy of a vaccine can be assessed with considerably smaller sample sizes and possibly more quickly, as compared to a conventional trial with community participants.9
Hasford still argues that31
…a challenge trial does not provide any direct benefit for the participant. A further important ethical issue is that since Hippocrates physicians have been asked to obey to two principles: beneficence and non-maleficence. Both are still indispensable and present an essential part of deontological ethics. Beneficence means that the physician has to take positive steps to contribute to the wellbeing of persons. The principle of nonmaleficence is often abbreviated to the words: ”Above all do no harm.” It is obvious that the intentional infection of persons, even if they are volunteers, means that physicians act in sharp contrast to these two principles that are still today part of the Hippocratic oath. The principal assumption of challenge trials is that harm has to be done to prevent greater harm for the society. There is no doubt, and the proponents of challenge trials admit too, that fatalities may occur in challenge trials.9
Hasford is among the many voices that have been speaking out about human challenge studies in the COVID-19 pandemic. Josh Morrison is also one of them. Morrison is not a scientist, but he felt the need to expand the impact of a possible vaccine against COVID-19 to as many people and countries as possible, when there was still no approved vaccine. The initiative led by Morrison, 1Day Sooner brings together healthy volunteers willing to be infected with the novel coronavirus to accelerate the development of vaccines and knowledge about COVID-19. Today, Morrison’s initiative has thousands of volunteers. As of April 4, 2021, the website indicated more than 38,000 people in 166 countries, according to data from the initiative. The idea of “altruistic motivation” has been linked to the motivations of volunteers.
It is important to emphasize that the use of challenge trials for research in humans is not new in the scientific community. Researchers have already used this model for research to develop vaccines against other diseases, such as malaria, dengue, and cholera.
However, as reflected in the criticism expressed by Hasford9, this fact does not make simple such a complex discussion about this type of study in the COVID-19 pandemic. In July 2020, a publication in Science drew attention to this problem. Entitled “Controversial ‘human challenge’ trials for COVID-19 vaccines gain support,”10 the publication indicates that the discussion on ethical issues related to these studies is far from over. In November 2020, weeks before the first vaccine was officially approved in the United Kingdom, a publication in the BMJ11 presented a counterpoint. While Josh Morrison, one of the authors, urges society to embrace the altruism of individuals who volunteer for this type of study, Charles Weijer, a bioethicist specialized in the study of ethics in clinical studies, considers the initiative dangerous and unjustified. Among the ethical concerns, the evaluation of the risks and benefits and the concern for the safety of these volunteers are highlighted.
Research ethics continues to play a crucial role
In this context, it may seem that there are no international ethical guidelines for human challenge trials focusing on COVID-19. However, the WHO has published guidelines for conducting this type of study. The “Key criteria for the ethical acceptability of COVID-19 human challenge studies” WHO document, of May 202012, provides guidance for scientists, Research Ethics Committees, funders, policy makers and regulators, in the deliberations on studies with human challenge in the fight against COVID-19. The document lists eight ethical criteria that must be considered for the approval of these studies.
According to that document12, for the stage of scientific and ethical assessments, the guidance is that these studies should have a strong scientific justification and the potential benefits should be expected to outweigh the risks; for the consultation and coordination stage, research programs must be informed by consultation and engagement by the public, as well as by experts and policy makers. They must also involve strong interaction among researchers, funders, policy makers and regulators. Regarding the selection criteria, human challenge trials must be carried out where the research can be conducted with the highest scientific and ethical standards. Such research must ensure that the selection criteria of the participants limit and minimize the risks; for the review and consent stage, the orientation is that the studies are reviewed by an independent specialized committee and involve strict informed consent from the participants, which we call Free and Informed Consent in Brazil.
However, in general, the regulatory aspects, in the various countries, mostly comprise conventional clinical trials.13 For example, if we consider the panorama of governmental policies for conducting clinical studies, the “ClinRegs”39 is an informative database that gathers international information on the regulation of clinical research. This database does not provide details on the regulation of human challenge trials.14 The database is associated with the National Institute of Allergy and Infectious Diseases (NIAID) of the United States, which publishes public policies on clinical research adopted by several countries.
In the United States, for example, regulatory aspects of clinical research can be identified in the work of the FDA (Food and Drug Administration). The FDA is associated with the Department of Health and Human Services and has a very important role in regulating clinical research. As described in the document “Conducting Clinical Studies”,15 the FDA is committed to “protecting participants in clinical studies, as well as providing reliable information to those interested in participating in studies.” The FDA establishes guidelines for clinical research that, in addition to those of other regulatory bodies, such as the Institutional Review Boards (IRBs), contribute to protect research participants from unjustified risks:
Under FDA regulations, an Institutional Review Board is group that has been formally designated to review and monitor biomedical research involving human subjects. In accordance with FDA regulations, an IRB has the authority to approve, require modifications in (to secure approval), or disapprove research. This group review serves an important role in the protection of the rights and welfare of human research subjects. The purpose of IRB review is to assure, both in advance and by periodic review, that appropriate steps are taken to protect the rights and welfare of humans participating as subjects in the research.16
In China, the State Drug Administration is the body that is responsible for the regulation, approval, and supervision of clinical studies. In the United Kingdom, the Medicines and Healthcare Products Regulatory Agency (MHRA) is responsible for this type of regulation of clinical research. Just as in the United States, with the role of IRBs, in both countries it is also necessary to analyze ethical aspects of the proposal, by the responsible bodies, for conducting clinical research.
As for Brazil, the National Health Surveillance Agency (ANVISA, Agência Nacional de Vigilância Sanitária) is the regulatory authority responsible for the approval, supervision, and inspection of clinical studies. ANVISA is an independent agency but linked to the Ministry of Health. Among the Agency’s various attributions, is the role of “inspecting Brazilian research centers to verify compliance to Best Clinical Practices in clinical studies”.17 ANVISA analyzes regulatory clinical studies and has been playing a major part in the approval of studies on COVID-19 in Brazil.
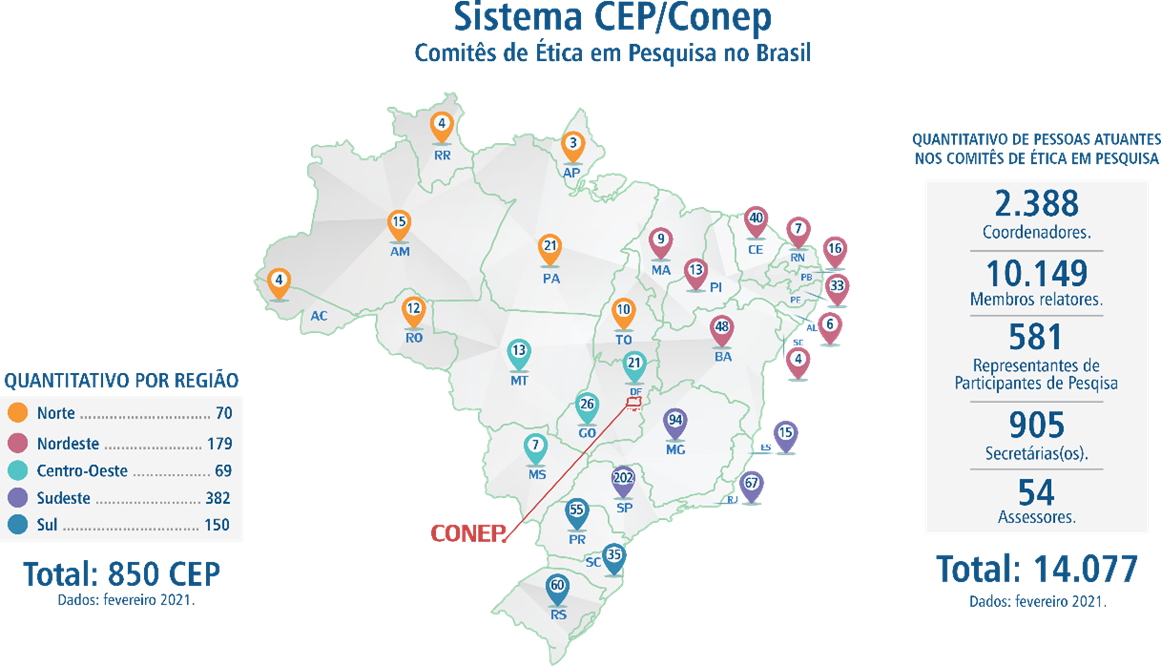
Regarding the ethical issues involved in these studies, the regulatory agencies rely on the important contribution of the Research Ethics Committees. In Brazil, the ethical regulation of research involving human beings, including clinical trials, is the responsibility of these Research Ethics Committees (CEP, Comitê de Ética em Pesquisa), which are distributed in all regions of the country. According to the CEP Map18, until February 2021, Brazil had 850 CEPs.
Figure 1: CEP Map.
CEPs “…. interdisciplinary and independent groups of a public function and advisory, deliberate, and educational nature, established to protect the interests of all research participants in their integrity and dignity and to contribute to the research development according to ethical standards.”19 Committees are linked to the National Research Ethics Commission (Comissão Nacional de Ética em Pesquisa), known as CONEP. Connected to the National Health Council (CNS, Conselho Nacional de Saúde), CONEP is responsible for preparing and updating the norms that protect the individual who participates in research.
Among these rules, the CNS Resolution 466 of 2012 stands out. CNS Resolution 466/12 “…seeks to ensure the rights and duties that refer to the participants of the research, to the scientific community and the State,”20 as described in its preliminary provisions. Resolution 466 has a fundamental role in the ethical regulation of clinical research. It establishes that every participant needs to be informed about the objectives, methods, possible benefits, and potential risks involved in their participation. In Brazil, payment of any kind to participants is forbidden, except in cases specified in the Resolution.
CONEP, when preparing and updating guidelines for the protection of research participants, considers that research involving human beings and ethical issues are inextricably linked. In the ethical evaluation of proposed projects, the CEPs consider, for example, the clarity of the description of risks and benefits, both known and potential, for participants in a given study. Among the ethical aspects observed by the CEPs are those related to vulnerability. Both CEPs and researchers must be careful that the proposed study does not accentuate situations of vulnerability for the participants and for the communities involved.
In general, regarding the ethical assessment of projects that intend to involve human beings in research, the CEPs have an important social mission. These Committees contribute, in a decisive way, to guarantee the safety of the participants, with special attention to these individuals’ rights and dignity. In the field of ethical regulation of research in humans, although Brazil, through the CEP/CONEP System, has particular characteristics, the country shares international ethical principles. For the CEP/CONEP System, among the features of an ethical study involving humans is the guarantee that the willingness of research participants to contribute and remain or not in the study is freely and clearly expressed.
Concerning research in humans21 in the COVID-19 pandemic, research ethics continues to play a crucial role. Regarding human challenge trials, the ongoing international debate has been modulated and enriched by ethical considerations that should be addressed with the perspective of different actors, such as researchers, members of Research Ethics Committees and the public, in different countries. In this changing landscape, in which “COVID-19 turned the world upside down,”1 research ethics continues to play a marked role to deepen reflections and guide decisions that are made at the science and society interface.
Watch the video “Aspectos éticos e regulatórios sobre a pesquisa em seres humanos na pandemia de COVID-19” produced by the Research Ethics, Science Communication and Society Laboratory (LECCS) of the Institute of Medical Biochemistry Leopoldo de Meis (IBqM)/UFRJ.
A version of this same content is available in the video below, with accessibility elements.
Acknowledgments
We wish to thank the graduate students Cleide Lima and Fernanda Victorio, from the Professional Masters Program in Science Education (IBqM/UFRJ) and Marlon Machado, from the Biological Chemistry Program – Science Education area – (IBqM/UFRJ) for their collaboration in the preparation of the video associated to this publication.
Notes
*Programa de Química Biológica – área de concentração Educação, Gestão e Difusão em Biociências – Instituto de Bioquímica Médica Leopoldo de Meis (IBqM), Universidade Federal do Rio de Janeiro (UFRJ), Brasil.
**Programa de Mestrado Profissional em Educação, Gestão e Difusão em Biociências, IBqM/UFRJ.
***Colégio Pedro II
1. Committee for the Coordination of Statistical Activities. How COVID-19 is changing the world: a statistical perspective [online]. United Nations Statistical Commission. 2020 [viewed 28 April 2021]. Available from: https://unstats.un.org/unsd/ccsa/documents/covid19-report-ccsa.pdf
2. Lei no. 13.979, de 6 de fevereiro de 2020 [online]. Diário Oficial da União. 2020 [viewed 28 April 2021]. Available from: https://www.in.gov.br/en/web/dou/-/lei-n-13.979-de-6-de-fevereiro-de-2020-242078735
3. Folha informativa sobre COVID-19 | Organização Pan-Americana da Saúde: https://www.paho.org/pt/covid19
4. PAN AMERICAM HEALTH ORGANIZATION. Frequently Asked Questions about COVID-19 Candidate Vaccines and Access Mechanisms. [online]. Institutional Repository for Information Sharing (IRIS). 2021 [viewed 28 April 2021]. Available from: https://iris.paho.org/handle/10665.2/53195
5. COVID-19 clinical trials have increased by 639% with the US leading the way [online]. Global Data. 2021 [viewed 28 April 2021]. Available from: https://www.globaldata.com/covid-19-clinical-trials-increased-639-us-leading-way/
6. EYAL, N., CAPLAN, A. and PLOTKIN, S. Human challenge trials of COVID-19 vaccines still have much to teach us [online]. The BJM Opinion, 2021 [viewed 28 April 2021]. Available from: https://blogs.bmj.com/bmj/2021/01/08/human-challenge-trials-of-covid-19-vaccines-still-have-much-to-teach-us/
7. Ensaios clínicos [online]. Instituto Butantan [viewed 28 April 2021]. Available from: https://butantan.gov.br/pesquisa/ensaios-clinicos
8. JAMROZIK, E., et al. Key criteria for the ethical acceptability of COVID-19 human challenge studies: Report of a WHO Working Group. Vaccine [online]. 2021, vol. 39, no. 4, pp. 633-640 [viewed 28 April 2021]. https://doi.org/10.1016/j.vaccine.2020.10.075. Available from: https://bit.ly/3xy3E9e
9. HASFORD, J. Covid-19: Human challenge studies will see people purposefully infected with virus. BMJ [online]. 2020, m4101 [viewed 28 April 2021]. https://doi.org/10.1136/bmj.m4101. Available from: https://www.bmj.com/lookup/doi/10.1136/bmj.m4101
10. COHEN, J. Controversial ‘human challenge’ trials for COVID-19 vaccines gain support [online]. Science. 2020 [viewed 28 April 2021]. http://doi.org/10.1126/science.abd9203. Available from: https://www.sciencemag.org/news/2020/07/controversial-human-challenge-trials-covid-19-vaccines-gain-support
11. McPARTLIN, S.O., et al. COVID-19 vaccines: Should we allow human challenge studies to infect healthy volunteers with SARS-CoV-2? BMJ [online]. 2020, m4258 [viewed 28 April 2021]. https://doi.org/10.1136/bmj.m4258. Available from: https://www.bmj.com/lookup/doi/10.1136/bmj.m4258
12. WHO. Key criteria for the ethical acceptability of COVID-19 human challenge studies [online]. World Health Organization. 2020 [viewed 28 April 2021]. Available from: https://www.who.int/ethics/publications/key-criteria-ethical-acceptability-of-covid-19-human-challenge/en
13. In February, a press release on GOV.UK announced the first human challenge study on COVID-19, officially approved in the UK (also approved by the ethics body). As described in “up to 90 volunteers aged 18-30 years will be exposed to Covid-19 in a safe and controlled environment to increase understanding of how the virus affects people.”
14. Mentions to “challenge trials”, “human challenge studies” in ClinRegs could not be found for any of the countries listed on the platform as of April 5th, 2021.
15. Conducting Clinical Trials [online]. U.S. Food & Drug Administration. 2020 [viewed 28 April 2021]. Available from: https://www.fda.gov/drugs/development-approval-process-drugs/conducting-clinical-trials
16. Institutional Review Boards (IRBs) and Protection of Human Subjects in Clinical Trials [online]. U.S. Food & Drug Administration. 2019 [viewed 28 April 2021]. Available from: https://www.fda.gov/about-fda/center-drug-evaluation-and-research-cder/institutional-review-boards-irbs-and-protection-human-subjects-clinical-trials
17. Ensaios Clínicos [online]. Anvisa [viewed 28 April 2021]. Available from: https://bit.ly/3u28nxJ
18. COMISSÃO NACIONAL DE ÉTICA EM PESQUISA. Cartilha dos Direitos dos Participantes de Pesquisa [online]. 2020 [viewed 28 April 2021]. Available from: http://gg.gg/l70q5
19. Resolution No 466, of 12 December 2012 [online]. Conselho Nacional de Saúde. 2012 [viewed 28 April 2021]. Available from: https://bvsms.saude.gov.br/bvs/saudelegis/cns/2013/res0466_12_12_2012.html
20. Comissão Nacional de Ética em Pesquisa [online]. Conselho Nacional de Saúde [viewed 28 April 2021]. Available from: http://conselho.saude.gov.br/comissoes-cns/conep
21. As it can be noted, the focus of this perspective is ethics on research involving human beings, due to the nature of the research addressed. Ethics in research involving human beings, with methodologies typical of the humanities and social sciences, has a number of specificities, not explored in this contribution and in the associated video. In Brazil, the main Resolution of the National Health Council (CNS), with guidelines for research involving human beings, is 510/2016.
References
CALLAWAY, E. Dozens to be deliberately infected with coronavirus in UK ‘human challenge’ trials. Nature [online]. 2020, vol. 586, no. 7831, pp. 651-652 [viewed 28 April 2021]. https://doi.org/10.1038/d41586-020-02821-4. Available from: https://www.nature.com/articles/d41586-020-02821-4
COHEN, J. Controversial ‘human challenge’ trials for COVID-19 vaccines gain support [online]. Science. 2020 [viewed 28 April 2021]. http://doi.org/10.1126/science.abd9203. Available from: https://www.sciencemag.org/news/2020/07/controversial-human-challenge-trials-covid-19-vaccines-gain-support
COHEN, J. Studies that intentionally infect people with disease-causing bugs are on the rise [online]. Science. 2016 [viewed 28 April 2021]. http://doi.org/10.1126/science.aaf5726. Available from: https://www.sciencemag.org/news/2016/05/studies-intentionally-infect-people-disease-causing-bugs-are-rise
Comissão Nacional de Ética em Pesquisa [online]. Conselho Nacional de Saúde [viewed 28 April 2021]. Available from: http://conselho.saude.gov.br/comissoes-cns/conep
COMISSÃO NACIONAL DE ÉTICA EM PESQUISA. Cartilha dos Direitos dos Participantes de Pesquisa [online]. 2020 [viewed 28 April 2021]. Available from: http://gg.gg/l70q5
Committee for the Coordination of Statistical Activities. How COVID-19 is changing the world: a statistical perspective [online]. United Nations Statistical Commission. 2020 [viewed 28 April 2021]. Available from: https://unstats.un.org/unsd/ccsa/documents/covid19-report-ccsa.pdf
Conducting Clinical Trials [online]. U.S. Food & Drug Administration. 2020 [viewed 28 April 2021]. Available from: https://www.fda.gov/drugs/development-approval-process-drugs/conducting-clinical-trials
COVID-19 clinical trials have increased by 639% with the US leading the way [online]. Global Data. 2021 [viewed 28 April 2021]. Available from: https://www.globaldata.com/covid-19-clinical-trials-increased-639-us-leading-way/
CRODA, J.H.R. AND GARCIA, L.P. Resposta imediata da Vigilância em Saúde à epidemia da COVID-19. Epidemiol. Serv. Saúde [online]. 2020, vol. 29, no. 1, e2020002 [viewed 28 April 2021]. https://doi.org/10.5123/s1679-49742020000100021. Available from: http://ref.scielo.org/rmzvjn
Ensaios Clínicos [online]. Anvisa [viewed 28 April 2021]. Available from: https://bit.ly/3u28nxJ
Ensaios clínicos [online]. Instituto Butantan [viewed 28 April 2021]. Available from: https://butantan.gov.br/pesquisa/ensaios-clinicos
Ensaios clínicos com medicamentos aprovados para prevenção ou tratamento da COVID-19 [online]. Agência Nacional de Vigilância Sanitária [viewed 28 April 2021]. Available from: http://antigo.anvisa.gov.br/estudos-clinicos-covid-19
Ensaios clínicos em andamento [online]. Portal Fiocruz. 2021 [viewed 28 April 2021]. Available from: https://portal.fiocruz.br/vacina-covid-19-ensaios-clinicos
EYAL, N. Why Challenge Trials of SARS‐CoV‐2 Vaccines could be ethical despite risk of severe adverse events. Ethics Hum Res [online]. 2020, vol. 42, no. 4, pp. 24–34 [viewed 28 April 2021]. https://doi.org/10.1002/eahr.500056. Available from: https://onlinelibrary.wiley.com/doi/abs/10.1002/eahr.500056
EYAL, N., CAPLAN, A. and PLOTKIN, S. Human challenge trials of COVID-19 vaccines still have much to teach us [online]. The BJM Opinion, 2021 [viewed 28 April 2021]. Available from: https://blogs.bmj.com/bmj/2021/01/08/human-challenge-trials-of-covid-19-vaccines-still-have-much-to-teach-us/
HASFORD, J. Covid-19: Human challenge studies will see people purposefully infected with virus. BMJ [online]. 2020, m4101 [viewed 28 April 2021]. https://doi.org/10.1136/bmj.m4101. Available from: https://www.bmj.com/lookup/doi/10.1136/bmj.m4101
Human Coronavirus Types [online]. Centers for Disease Control and Prevention. 2020 [viewed 28 April 2021]. Available from: https://www.cdc.gov/coronavirus/types.html
Institutional Review Boards (IRBs) and Protection of Human Subjects in Clinical Trials [online]. U.S. Food & Drug Administration. 2019 [viewed 28 April 2021]. Available from: https://www.fda.gov/about-fda/center-drug-evaluation-and-research-cder/institutional-review-boards-irbs-and-protection-human-subjects-clinical-trials
JAMROZIK, E. and SELGELID, M.J. COVID-19 human challenge studies: ethical issues. Lancet Infect Dis [online]. 2020, vol. 20, no. 8, e198–203 [viewed 28 April 2021]. https://doi.org/10.1016/S1473-3099(20)30438-2. Available from: https://bit.ly/339lFwV
JAMROZIK, E., et al. Key criteria for the ethical acceptability of COVID-19 human challenge studies: Report of a WHO Working Group. Vaccine [online]. 2021, vol. 39, no. 4, pp. 633-640 [viewed 28 April 2021]. https://doi.org/10.1016/j.vaccine.2020.10.075. Available from: https://bit.ly/3xy3E9e
JOKURA, T. Distribution challenges [online]. Pesquisa FAPESP. 2021 [viewed 28 April 2021]. Available from: https://revistapesquisa.fapesp.br/en/distribution-challenges/
Lei no. 13.979, de 6 de fevereiro de 2020 [online]. Diário Oficial da União. 2020 [viewed 28 April 2021]. Available from: https://www.in.gov.br/en/web/dou/-/lei-n-13.979-de-6-de-fevereiro-de-2020-242078735
LEWIS, P. COVID-19: Health literacy and public health information [online]. House of Lords Library, 2020 [viewed 28 April 2021]. Available from: https://lordslibrary.parliament.uk/covid-19-health-literacy-and-public-health-information/
Listings of WHO’s response to COVID-19 [online]. World Health Organization. 2020 [viewed 28 April 2021]. Available from: https://www.who.int/news/item/29-06-2020-covidtimeline
MARON, D.F. “Wet markets” likely launched the coronavirus. Here’s what you need to know. [online]. National Geographic. 2020 [viewed 28 April 2021]. Available from: https://www.nationalgeographic.com/animals/article/coronavirus-linked-to-chinese-wet-markets
McPARTLIN, S.O., et al. COVID-19 vaccines: Should we allow human challenge studies to infect healthy volunteers with SARS-CoV-2? BMJ [online]. 2020, m4258 [viewed 28 April 2021]. https://doi.org/10.1136/bmj.m4258. Available from: https://www.bmj.com/lookup/doi/10.1136/bmj.m4258
MURRAY, C.J.L., et al. Digital public health and COVID-19. The Lancet Public Health [online]. 2020, vol. 5, no. 9, e469–e470 [viewed 28 April 2021]. https://doi.org/10.1016/S2468-2667(20)30187-0. Available from: https://bit.ly/3xvacW5
Naming the coronavirus disease (COVID-19) and the virus that causes it [online]. World Health Organization. 2020 [viewed 28 April 2021]. Available from: https://www.who.int/emergencies/diseases/novel-coronavirus-2019/technical-guidance/naming-the-coronavirus-disease-(covid-2019)-and-the-virus-that-causes-it
PAAKKARI, L. and OKAN, O. COVID-19: Health literacy is an underestimated problem. The Lancet Public Health [online]. 2020, vol. 5, no. 5, e249-250 [viewed 28 April 2021]. https://doi.org/10.1016/S2468-2667(20)30086-4. Available from: https://www.ncbi.nlm.nih.gov/pmc/articles/PMC7156243/
PAN AMERICAM HEALTH ORGANIZATION. Frequently Asked Questions about COVID-19 Candidate Vaccines and Access Mechanisms. [online]. Institutional Repository for Information Sharing (IRIS). 2021 [viewed 28 April 2021]. Available from: https://iris.paho.org/handle/10665.2/53195
PARKER, L. To find a vaccine for COVID-19, will we have to deliberately infect people? [online]. National Geographic. 2020 [viewed 28 April 2021]. Available from: https://www.nationalgeographic.co.uk/science-and-technology/2020/09/to-find-a-vaccine-for-covid-19-will-we-have-to-deliberately-infect
Resolution No 466, of 12 December 2012 [online]. Conselho Nacional de Saúde. 2012 [viewed 28 April 2021]. Available from: https://bvsms.saude.gov.br/bvs/saudelegis/cns/2013/res0466_12_12_2012.html
RUBIN, R. Challenge Trials—Could Deliberate Coronavirus Exposure Hasten Vaccine Development? JAMA [online]. 2020, vol. 324, no. 1, pp. 12-14 [viewed 28 April 2021]. http://doi.org/10.1001/jama.2020.9881. Available from: https://jamanetwork.com/journals/jama/fullarticle/2767024/
Timeline: WHO’s COVID-19 response [online]. World Health Organization. 2021 [viewed 28 April 2021]. Available from: https://bit.ly/32YpPHA
UNITED NATIONS DEPARTMENT OF GLOBAL COMMUNICATIONS. 5 ways the UN is fighting ‘infodemic’ of misinformation [online]. United Nations. 2020 [viewed 28 April 2021]. Available from: https://www.un.org/en/un-coronavirus-communications-team/five-ways-united-nations-fighting-‘infodemic’-misinformation
Update on COVID-19 vaccine development [online]. World Health Organization. 2020 [viewed 28 April 2021]. Available from: https://www.who.int/docs/default-source/coronaviruse/risk-comms-updates/update45-vaccines-developement.pdf?sfvrsn=13098bfc_5
WHO calls for further studies, data on origin of SARS-CoV-2 virus, reiterates that all hypotheses remain open [online]. World Health Organization. 2021 [viewed 28 April 2021]. Available from: https://www.who.int/news/item/30-03-2021-who-calls-for-further-studies-data-on-origin-of-sars-cov-2-virus-reiterates-that-all-hypotheses-remain-open
WHO Director-General’s opening remarks at the media briefing on COVID-19 – 11 March 2020 [online]. World Health Organization. 2020 [viewed 28 April 2021]. Available from: https://www.who.int/director-general/speeches/detail/who-director-general-s-opening-remarks-at-the-media-briefing-on-covid-19—11-march-2020
WHO. Key criteria for the ethical acceptability of COVID-19 human challenge studies [online]. World Health Organization. 2020 [viewed 28 April 2021]. Available from: https://www.who.int/ethics/publications/key-criteria-ethical-acceptability-of-covid-19-human-challenge/en
World Health Organization (WHO) [social network]. In: @WHO [online]. Twitter, March 11, 2020 [viewed 28 April 2021]. Available from: https://twitter.com/WHO/status/1237777021742338049
YAN, W. Challenge accepted: Human challenge trials for dengue. Nat Med [online]. 2015, vol. 21, no. 8, pp. 828–830 [viewed 28 April 2021]. https://doi.org/10.1038/nm0815-828. Available from: http://www.nature.com/articles/nm0815-828
External links
1Day Sooner: https://www.1daysooner.org/
Agência Nacional de Vigilância Sanitária – Anvisa: https://www.gov.br/anvisa/pt-br
ClinRegs: https://clinregs.niaid.nih.gov/
Conselho Nacional de Saúde – Mapa dos CEPs: http://conselho.saude.gov.br/contato-cns/93-comissoes/conep/comites-de-etica-em-pesquisa-conep/645-mapa-dos-ceps-conep
Folha informativa sobre COVID-19 | Organização Pan-Americana da Saúde: https://www.paho.org/pt/covid19
Linha do tempo coronavírus: https://coronavirus.saude.gov.br/linha-do-tempo/
WHO Coronavirus (COVID-19) Dashboard | WHO Coronavirus (COVID-19) Dashboard With Vaccination Data: https://covid19.who.int/
Translated from the original in Portuguese by Lilian Nassi-Calò.
[Revised on April 25, 2022]
Como citar este post [ISO 690/2010]:
















Recent Comments